An international team of researchers headed by Kensuke Kimura of the Surface and Interface Science Laboratory, RIKEN (Wako, Japan) is developing a technology that has the potential to enable lower energy consumption in OLED displays.
A recent publication by the team is entitled “Selective triplet exciton formation in a single molecule.” It was published in Nature volume 570, pages 210-213 (2019). A copy of the article is available for purchase here.
To start, here are a few words of background information.
Electrical current passing through an OLED material causes the formation of excitons. Excitons are pairs of electrons and holes. More specifically, current injected into an OLED material forms spin-singlet excitons (having opposite spins) and spin-triplet excitons (having the same spins) in the ratio of 1 to 3. Singlets are the higher energy state and can convert into triplets. When either a singlet or a triplet converts to a lower energy state, light is emitted.
Phosphorescence in the current generation of OLED materials is typically based on the decay of triplets. Based on this fact, the use of energy to create higher energy singlets is not efficient. A material/process that creates only triplets and that produces light exclusive by the means of triplet decay should have a lower OLED operating voltage and, thus, enable an improved overall OLED display energy efficiency. At this time, however, effective means to enhance direct formation of triplets has not been developed. The research undertaken by the team was intended to address this issue.
The team’s most recent work began with research to understand the basic physics behind the creation of excitons. To this end, the team prepared a model system based on an organic semiconductor called 3,4,9,10-perylenetetracarboxylic dianhydride (PTCDA). This semiconductor was adsorbed on an ultrathin insulating film composed of three monolayers of NaCl that was, in turn, supported by a metal film composed of Ag(111). (Note that the (111) notation refers to the crystal structure of silver.) A technique described in their article was used to impart a negative charge to the molecule. In the next step, current from a Scanning Tunneling Microscope (STM) was used to induce luminescence in the molecule. The type of exciton created by this process was determined by an optical detection system that analyzed the spectrum of the emitted light.
Both phosphorescence and fluorescence signals where found when a high voltage was applied. In contrast, only phosphorescence was found to occur when a low voltage was applied. These results indicate the selective formation of triplet excitons without creating their singlet counterparts. Theoretical calculations confirmed the experimental results and verified the mechanism.
Part (a) of the figure below illustrates the configuration of the measurement. Part (b) is a scanning tunneling microscope image of PTCDA adsorbed on NaCl grown on Ag(111). Part (c) is the luminescence spectrum produced by the material.
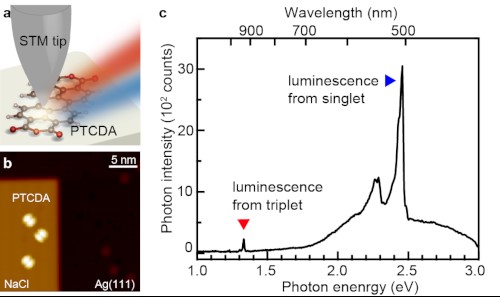 (a) Schematic image of the measurement, (b) Scanning tunneling microscope image of PTCDA adsorbed on NaCl grown on Ag(111), (c) Luminescence spectrum.
(a) Schematic image of the measurement, (b) Scanning tunneling microscope image of PTCDA adsorbed on NaCl grown on Ag(111), (c) Luminescence spectrum.
In describing the experimental results reported in their article, the team stated that
“We believe that we were able to do this thanks to a previously unknown mechanism, where electrons are selectively removed from the charged molecule depending on their spin state.”
They go on to explain that
“a new way to manipulate the excitons was demonstrated that is essential to the transport of electrons within OLEDs. That the electron transport process accompanying exciton formation in OLEDs can be controlled by manipulating an electron spin inside a molecule.”
The article concludes with the speculation that “designing a device taking into account the exchange interaction could realize an OLED with a lower operating voltage.” -Arthur Berman
